Aim
This software is aimed to facilitate research collaborations between a network of clinical and technical partners that is needed to address the challenge of cost effective and efficient lung CT screening. A radiologist can quickly and accurately report chest CT scans and produce a standardized report with CIRRUS Lung Screening according to Lung-RADS guidelines. AI algorithms developed within DIAG are integrated into the workstation to allow for more accurate and faster interpretation of CT scans and extraction of a rich set of biomarkers. In addition to screen for lung cancer, the workstation is also aimed to screen for two other conditions for which heavy smokers are at risk: chronic obstructive pulmonary disease (COPD) and cardiovascular diseases.
Software
This software includes computer-aided detection and volumetric
measurements for both solid and subsolid nodules. The workflow can be
categorized into three steps - processing, reading and reporting.
During the processing stage, input DICOM data is validated first. Then,
lung, airway and vessel, lobe and segments are segmented. Various CAD
systems are run to detect nodules, which is then followed by a
dedicated lung CT registration algorithm to register a new scan with
prior scans. All nodules from the prior scans are propagated
automatically to the new scan. Finally, the prior findings and the CAD
marks are merged. This software also provides a functionality to
propagate a newly found nodule retroactively through the previous scans.
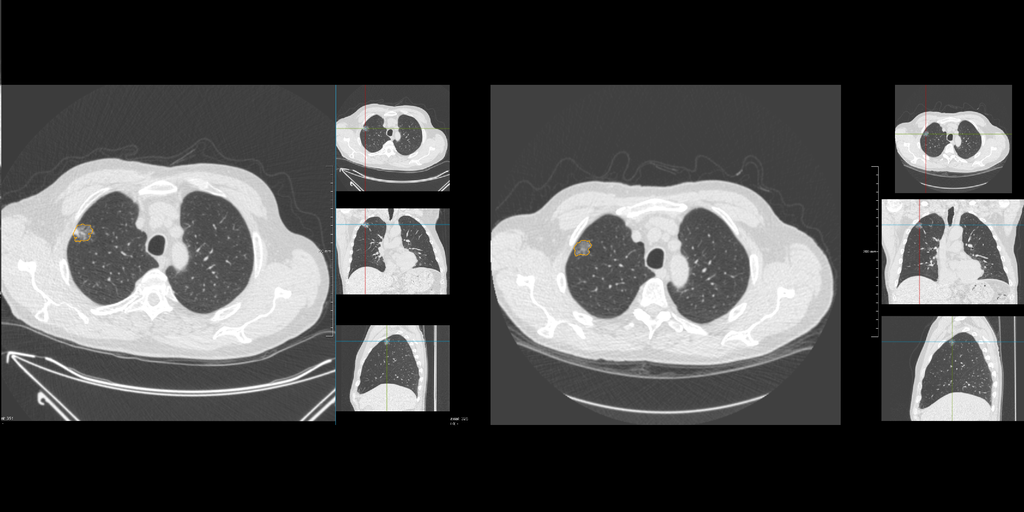 CIRRUSLungS workstation
CIRRUSLungS workstation
After a user has finished reading, a structured report is automatically
generated. The report provides general patient information, gives a
quick overview of the annotated findings and their characteristics and
optionally provides follow-up recommendations according to the
BTS/Fleischner/LungRADS guidelines. The suggested follow up and other
case comments can be added to the report. Finally, the user signs off
the case and proceeds with the next one.
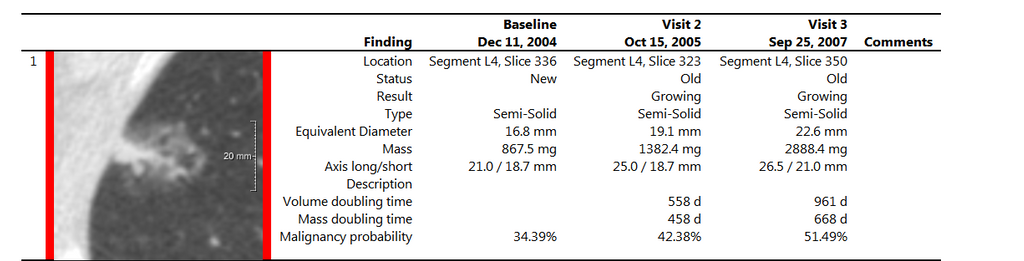 CIRRUSLungS report
CIRRUSLungS report
Usage
There are two different versions of our software. The research version runs the processing pipeline on our in-house clusters and contains experimental features. The commercial version, Veolity, uses a client-server architecture, has communication with PACS systems integrated, can use PACS as a storage database, divides worklists over multiple clients, and is CE and FDA certified. Both versions are in active use at sites in North America, Europe, Asia and Australia.



